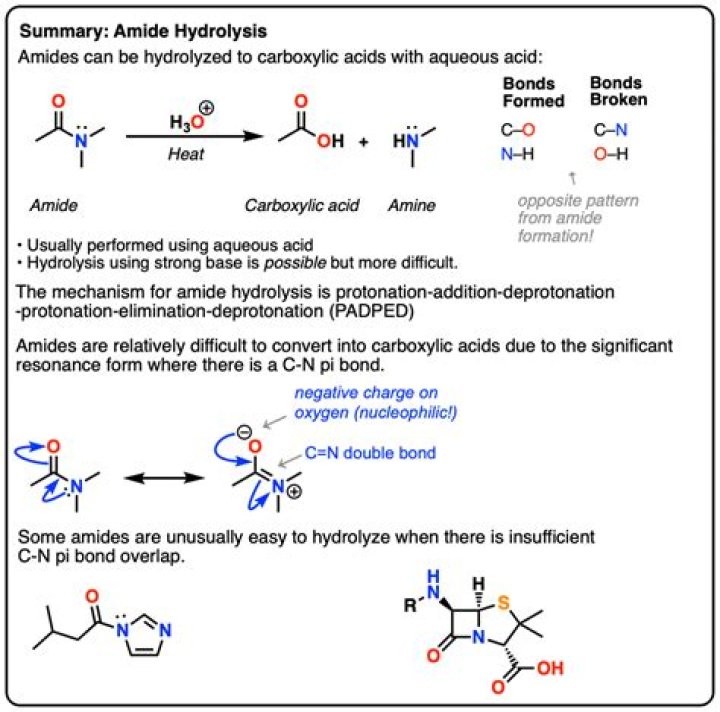
Hydrolysis of an amide in acid solution actually gives a carboxylic acid and the salt of ammonia or an amine (the ammonia or amine initially formed is neutralized by the acid). Basic hydrolysis gives a salt of the carboxylic acid and ammonia or an amine.
What happens when amide is hydrolysed?
The mechanism involves protonation of the amide on oxygen followed by attack of water on the carbonyl carbon. The tetrahedral intermediate formed dissociates ultimately to the carboxylic acid and the ammonium salt: In alkaline hydrolysis the amide is heated with boiling aqueous sodium or potassium hydroxide.
Can amides undergo hydrolysis?
Amides can also undergo hydrolysis either under acidic or basic conditions to produce carboxylic acids. Under basic conditions, the hydroxide acts as the nucleophile, reacting with the amide to form an intermediate anion.
What does HCl do to an amide?
In the presence of a strong acid, such as HCl, an amide reacts with water to produce a carboxylic acid and an ammonium or amine salt. In the presence of a strong base, like NaOH, the amide reacts to produce ammonia or an amine, and a carboxylic acid salt.
What reaction occurs after an amide is hydrolyzed?
Hydrolysis of an amide breaks the carbon–nitrogen bond and produces a carboxylic acid and either ammonia or an amine.
What is acid hydrolysis biochemistry?
In organic chemistry, acid hydrolysis is a hydrolysis process in which a protic acid is used to catalyze the cleavage of a chemical bond via a nucleophilic substitution reaction, with the addition of the elements of water (H2O). For example, in the conversion of cellulose or starch to glucose.
Is acid hydrolysis of amides reversible?
Esters and amides are two of the least reactive carboxylic acid derivatives. Acid-catalysed hydrolysis reactions are reversible. The forward reaction is driven over to product by using an excess of water, usually as the solvent.
Why is the hydrolysis of amides irreversible?
Acid-catalysed hydrolysis reactions are reversible. The forward reaction is driven over to product by using an excess of water, usually as the solvent. Base-mediated hydrolyses, however are essentially irreversible because the product is a non-electrophilic carboxylate salt.
How do amides react with water in an acid solution?
Technically, hydrolysis is a reaction with water. That is exactly what happens when amides are hydrolyzed in the presence of dilute acids such as dilute hydrochloric acid. The acid acts as a catalyst for the reaction between the amide and water.
Why are amides so stable?
Amides are the most stable, and the least reactive, because nitrogen is an effective donor of electrons to the carbonyl group. Anhydrides are less stable because the donation of electrons to one carbonyl group is in competition with the donation of electrons to the second carbonyl group.