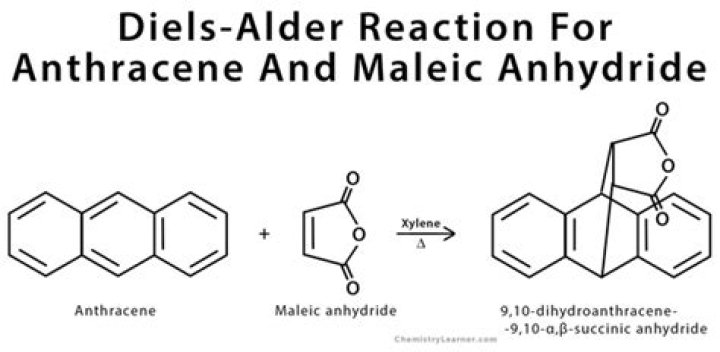
A particularly rapid Diels-Alder reaction takes place between cyclopentadiene and maleic anhydride. In general, Diels-Alder reactions proceed fastest with electron-donating groups on the diene (eg. alkyl groups) and electron-withdrawing groups on the dienophile.
When using maleic anhydride in a Diels-Alder reaction Why is it important that the glassware and reagents are dry?
1, Expt. 13. Since both maleic anhydride and the product of the Diels-Alder reaction can be hydrolyzed by water, it is important to use dry glassware and to exclude moisture during the reaction and the work- up. The melting point of the product will reveal the identity of the conjugated diene present in the oil.
Why is maleic anhydride a reactive dienophile in Diels-Alder reactions?
In terms of activation, notice that maleic anhydride is a highly reactive dienophile, due to the presence of two electron- withdrawing carbonyl substituents.
What is the best dienophile for a Diels-Alder reaction?
Maleic anhydride is also a very good dienophile, because the electron-withdrawing effect of the carbonyl groups causes the two alkene carbons to be electron-poor, and thus a good target for attack by the pi electrons in the diene.
What type of reaction is Diels-Alder?
In organic chemistry, the Diels–Alder reaction is a chemical reaction between a conjugated diene and a substituted alkene, commonly termed the dienophile, to form a substituted cyclohexene derivative. It is the prototypical example of a pericyclic reaction with a concerted mechanism.
What type of reaction is Diels Alder?
What action should you take if maleic anhydride gets in your eyes?( 3 pts?
exposures may cause a build-up of fluid in the lungs (pulmonary edema), a medical emergency. ► Exposure to Maleic Anhydride can cause headache, dizziness, nausea and vomiting. allergy.
Which of the following dienophile is the most reactive with Buta 1/3 diene?
The most reactive dienophile is the aldehyde — propenal.
Which acid can be used as a dienophile in cycloaddition reaction?
The aza Diels-Alder reaction involves the use of imines as the dienophile (or diene substituents).