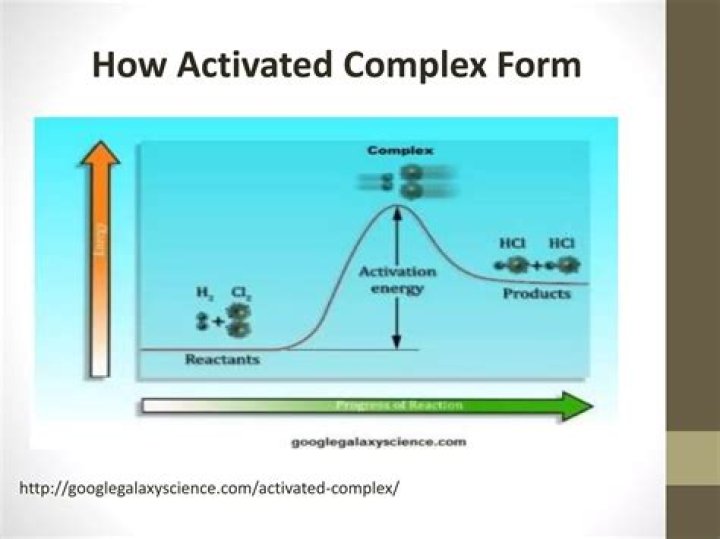
❖ Activated Complex Theory In 1935, an American chemist Henry Eyring; alongside two British chemists, Meredith Gwynne Evans and Michael Polanyi; proposed a new theory to rationalize the rate of different chemical reactions which was based upon the formation of an activated intermediate complex.
What is activated energy theory?
In transition-state theory, the activation energy is the difference in energy content between atoms or molecules in an activated or transition-state configuration and the corresponding atoms and molecules in their initial configuration. …
What does activated complex mean in chemistry?
An activated complex is an unstable arrangement of atoms that exists momentarily at the peak of the activation energy barrier. Because of its high energy, the activated complex exists for an extremely short period of time (about 10−13s).
What is activated complex example?
A new bond between H and F must also form to make HF. The reactant bonds break at the same time that the product bonds form. This is called the activated complex or transition state. In this example, the final product is HF and it has a lower energy than the reactants.
What is difference between transitional and activated complex?
The main difference between activated complex and transition state is that activate complex refers to all possible intermediates whereas transition state refers to the intermediate with the highest potential energy.
Why is the activated complex theory said to be superior to the collision theory?
Activated complex theory and collision theory are important thermodynamic theories. The key difference between activated complex theory and collision theory is that the activated complex theory is more accurate and reliable when describing the reaction rates, whereas collision theory is less reliable.
What is activation energy give example?
They require a certain amount of energy just to get started. This energy is called activation energy. For example, activation energy is needed to start a car engine. Turning the key causes a spark that activates the burning of gasoline in the engine.
What is activated complex quizlet?
Activated Complex. A short-lived, unstable arrangement of atoms that may break apart and re-form the reactants or may form products; also sometimes referred to as the transition state.
What is the role of the activated complex?
The term activated complex refers to the molecular compound or compounds that exist in the highest energy state, or activated stage, during a chemical reaction. An activated complex acts as an intermediary between the reactants and the products of the reaction.
What is a reactant complex?
An activated complex is an intermediate state that is formed during the conversion of reactants into products. An activated complex is the structure that results in the maximum energy point along the reaction path.
What is the difference between activation energy and activated complex?
Activated complex is an intermediate structure formed in the conversion of reactants to products. The activated complex is the structure at the maximum energy point along the reaction path; the activation energy is the difference between the energies of the activated complex and the reactants.