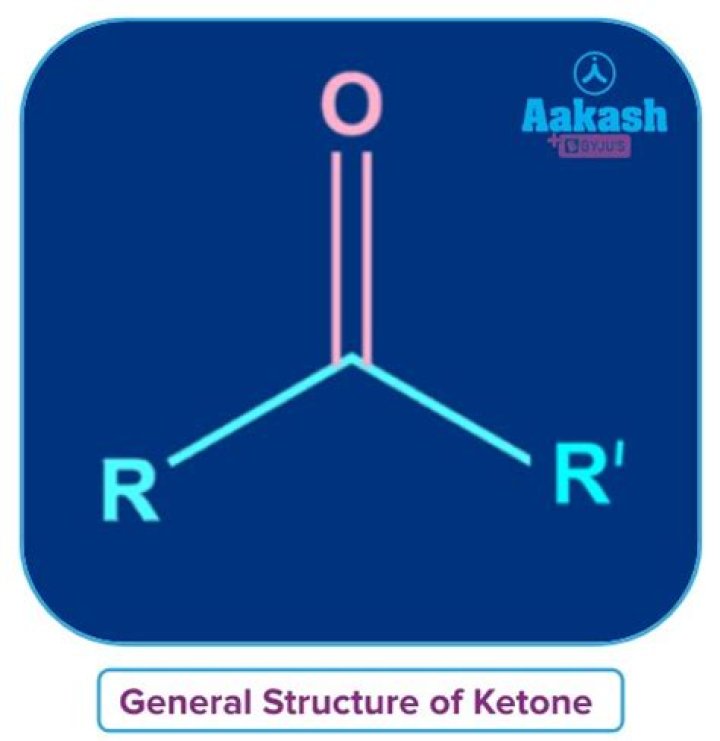
In chemistry, a ketone /ˈkiːtoʊn/ is a functional group with the structure R2C=O, where R can be a variety of carbon-containing substituents. Ketones contain a carbonyl group (a carbon-oxygen double bond). The simplest ketone is acetone (R = R’ = methyl), with the formula CH3C(O)CH3.
What are ketone groups?
ketone, any of a class of organic compounds characterized by the presence of a carbonyl group in which the carbon atom is covalently bonded to an oxygen atom. The remaining two bonds are to other carbon atoms or hydrocarbon radicals (R): Fast Facts.
What are the functional group of ketones?
Aldehydes and ketones are organic compounds which incorporate a carbonyl functional group, C=O. The carbon atom of this group has two remaining bonds that may be occupied by hydrogen or alkyl or aryl substituents.
Is ketone a carbonyl group?
Aldehydes and ketones contain the carbonyl group. Ketones contain the carbonyl group bonded to two carbon atoms. Aldehydes and ketones are organic compounds which incorporate a carbonyl functional group, C=O.
What is amide structure?
Amides have a general structure in which a nitrogen atom is bonded to a carbonyl carbon atom. The functional group for an amide is as follows: In names for amides, the -ic acid of the common name or the -oic ending of the IUPAC for the corresponding carboxylic acid is replaced by -amide.
How do you identify a ketone group?
Ketones are named the same way as are alkenes except that an -one ending is used. The location of the carbonyl group in the molecule is identified by numbering the longest chain of carbons so that the carbonyl group has the lowest number possible.
Do ketones contain carbonyl?
The carbonyl group (C=O) is found in aldehydes, ketones, and many other organic functional groups. The carbon and oxygen in the carbonyl group are sp2-hybridized, with bond angles of 120°.
Is 2 pentanone an aldehyde or ketone?
IUPAC Rules for Naming Ketones
| Propanone (acetone) | Butanone (methyl ethyl ketone) |
|---|---|
| 2-Pentanone | 3-Pentanone |
| Acetophenone | Benzophenone |
What’s the difference between amines and amides?
Compounds containing a nitrogen atom bonded in a hydrocarbon framework are classified as amines. Compounds that have a nitrogen atom bonded to one side of a carbonyl group are classified as amides.
What is the simplest amide?
Formamide
Formamide, the simplest amide.
What is a keto acid made of?
Keto acid. Keto acids or ketoacids (also called oxo acids or oxoacids) are organic compounds that contain a carboxylic acid group and a ketone group. In several cases, the keto group is hydrated.
What is the IUPAC nomenclature for ketone functional groups?
When the two alkyl groups are the same, the prefix is added before the name of alkyl group. The positions of other groups are indicated by Greek letters, the α-carbon being the atom adjacent to carbonyl group. Although used infrequently, oxo is the IUPAC nomenclature for a ketone functional group.
What is the difference between beta keto and gamma keto acids?
Beta-keto acids, Beta-ketoacids, or 3-oxoacids, such as acetoacetic acid, have the ketone group at the second carbon from the carboxylic acid. They generally form by the Claisen condensation. Gamma-keto acids, Gamma-ketoacids, or 4-oxoacids have the ketone group at the third carbon from the carboxylic acid. Levulinic acid is an example.
What is the difference between ketones and ketone bodies?
Not to be confused with ketone bodies. In chemistry, a ketone / ˈkiːtoʊn / is a functional group with the structure R 2 C=O, where R can be a variety of carbon-containing substituents. Ketones contain a carbonyl group (a carbon-oxygen double bond).