CaCO3
Calcium carbonate/Formula
Is CaCl2 a compound?
Calcium chloride
Calcium chloride/IUPAC ID
Calcium chloride is an inorganic compound, a salt with the chemical formula CaCl2. It is a white colored crystalline solid at room temperature, and it is highly soluble in water.
Which compound is represented by caco3?
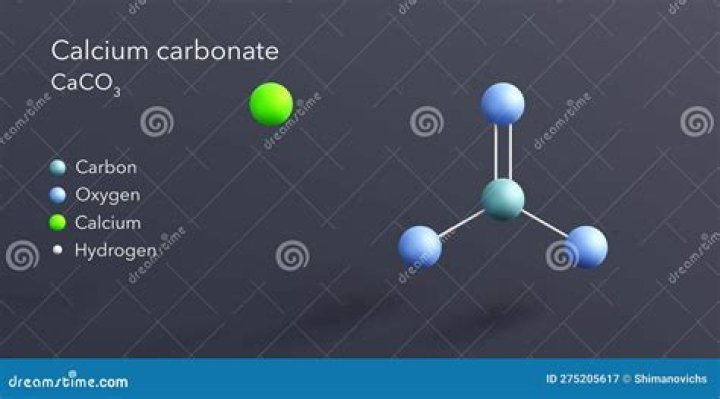
Calcium carbonate
Calcium carbonate is a chemical compound with the formula CaCO3 formed by three main elements: carbon, oxygen, and calcium.
How does calcium affect the growth of plants?
Calcium increases ammonium, potassium and phosphorus absorption, stimulates photosynthesis, and increases the size of sellable plant parts. It also makes the use of nitrogen more efficient, which improves the economics of production and reduces nitrogen contamination of the environment.
How do you solve caco3?
CaCO3 is obtained by using carbon dioxide and slaked lime as raw materials. When carbon dioxide is passed through slaked lime, calcite is obtained. Another method to obtain calcite is by adding sodium carbonate to calcium chloride.
What is the composition of calcium in caco3?
The percentage compositions of the elements of calcium carbonate are given as: Ca = 40%; C = 12%; O = 48%.
What are the compounds for cacl2?
The name for CaCl2 is calcium chloride. This is a binary ionic compound, a metal and a non metal.
How do you make cacl2?
CaCl2 stock solution To prepare a 2.5 M stock solution of CaCl2, dissolve 11 g of CaCl2•6H2O in a final volume of 20 mL of H2O. Sterilize the solution by passing it through a 0.22-μm filter. Store in 1-mL aliquots at 4°C.
What is the composition of calcium in CaCO3?
How do I give my plants calcium?
Adding lime to the soil in autumn is the easiest answer to how to raise calcium in the soil. Eggshells in your compost will also add calcium to soil. Some gardeners plant eggshells along with their tomato seedlings to add calcium to soil and prevent blossom end rot.
How do you measure calcium in plants?
Generally, Atomic Absorption Instruments (AA) or Inductivity Coupled Plasma-Optical Emission Spectrometry Instruments (ICP-OES) are used to measure the amount of calcium ions present in soil.