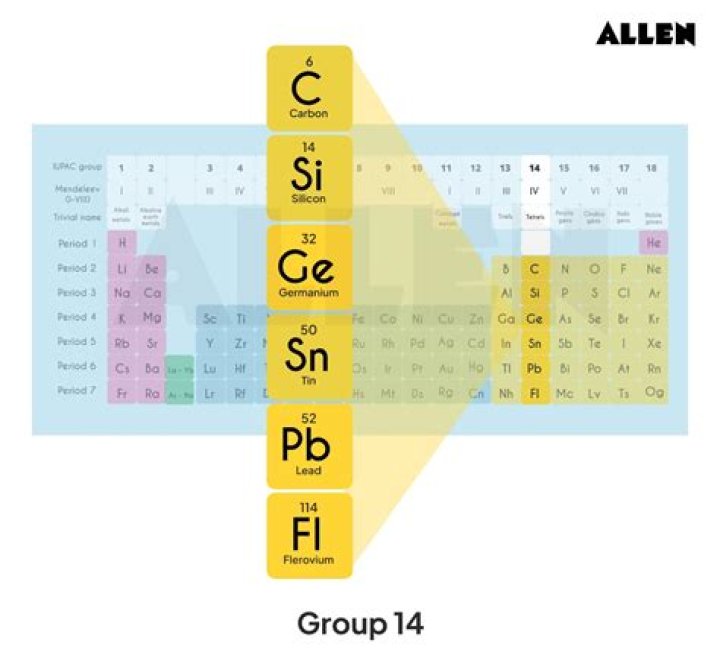
Group 14 is the carbon family. The five members are carbon, silicon, germanium, tin, and lead. All of these elements have four electrons in their outermost energy level. Of the Group 14 elements, only carbon and silicon form bonds as nonmetals (sharing electrons covalently).
What is called group 14?
The carbon group is a periodic table group consisting of carbon (C), silicon (Si), germanium (Ge), tin (Sn), lead (Pb), and flerovium (Fl). It lies within the p-block. In modern IUPAC notation, it is called group 14. They are also known as the crystallogens or adamantogens.
How do you remember the elements in group 14?
Group 14 is known as Carbon group or the group of Crystallogens, Tetragens or Tetrels. It includes Carbon (C), Silicon (Si), Germanium (Ge), Tin (Sn), and Lead (Pb). Mnemonic for Group 14: Chemistry Sir Gives Sanki Problems.
What charge do group 14 elements have?
And elements in group 14 have a charge of -4. Elements in group 16 have a charge of -2, while all the elements of group 17 are halogens with a charge of -1 each.
Why are group 14 elements called Crystallogens?
The carbon family is also called the carbon group, group 14, or group IV. At one time, this family was called the tetrels or tetragens because the elements belonged to group IV or as a reference to the four valence electrons of atoms of these elements. The family is also called the crystallogens.
Which of the following elements of group 14 have maximum allotropes?
Next to sulfur, carbon is the element with the most allotropes. Carbon has three main solid state allotropes: graphite, diamond, and fullerenes (the most commonly known of which, buckminsterfullerene, is also known as a “buckyball”).
How many metals are in group 14 on the periodic table?
two metals
Element density increases moving down the group. The carbon family consists of one nonmetal (carbon), two metalloids (silicon and germanium), and two metals (tin and lead).
Which compound contains a group 14 metal?
The carbon family, Group 14 in the p-block, contains carbon (C), silicon (Si), germanium (Ge), tin (Sn), lead (Pb), and flerovium (Fl)….Introduction.
| Element | Lead |
|---|---|
| Symbol | Pb |
| Atomic Mass | 207.2 |
| Classification | Metal |
| Electron Configuration | [Xe]4f145d106s26p2 |
Are carbon and silicon in the same group?
carbon group element, any of the six chemical elements that make up Group 14 (IVa) of the periodic table—namely, carbon (C), silicon (Si), germanium (Ge), tin (Sn), lead (Pb), and flerovium (Fl).