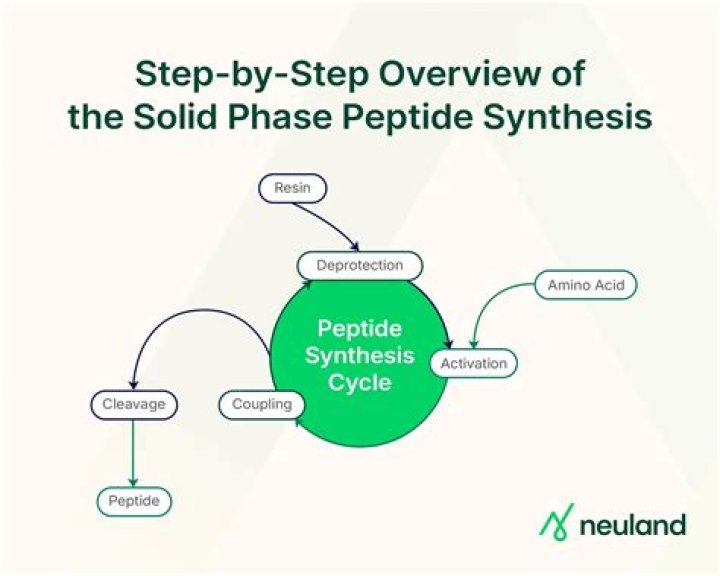
Solid-phase peptide synthesis (SPPS) involves the successive addition of protected amino acid derivatives to a growing peptide chain immobilized on a solid phase, including deprotection and washing steps to remove unreacted groups and also side products.
What is the process of peptide synthesis?
In organic chemistry, peptide synthesis is the production of peptides, compounds where multiple amino acids are linked via amide bonds, also known as peptide bonds. Peptides are chemically synthesized by the condensation reaction of the carboxyl group of one amino acid to the amino group of another.
How do you synthesize cyclic peptides?
Cyclic peptides can also be synthesized by linking the amino (N) terminus of the peptide to the carboxyl (C) terminus via an amide bond. The amino side chains of Lys and Orn and the carboxyl side chains of Asp and Glu can also be used to construct cyclic peptides via an amide bond.
What is solid-phase peptide synthesis used for?
This method is used for the synthesis of peptides, deoxyribonucleic acid (DNA), ribonucleic acid (RNA), and other molecules that need to be synthesised in a certain alignment. More recently, this method has also been used in combinatorial chemistry and other synthetic applications.
What are peptides explain solid-phase peptide synthesis?
The concept of solid-phase peptide synthesis (SPPS) is to retain chemistry that has been proven in solution but to add a covalent attachment step that links the nascent peptide chain to an insoluble polymeric support (resin). Subsequently, the anchored peptide is extended by a series of addition cycles (Fig. 18.1. 1).
How does solid phase peptide synthesis work?
Solid-phase synthesis is a common technique for peptide synthesis. In peptide synthesis, an amino-protected amino acid is bound to a solid phase material or resin (most commonly, low cross-linked polystyrene beads), forming a covalent bond between the carbonyl group and the resin, most often an amido or an ester bond.
What is the first step in solid phase peptide synthesis?
What are macrocyclic peptides?
Macrocyclic peptides are a remarkably diverse family of peptides found in a range of organisms, including bacteria, plants, fungi, and mammals. Nonnative residues, which can enhance biological stability, are often present in the smaller peptides, whereas the larger peptides tend to have predominantly standard residues.
Which of the following peptides is cyclic in nature -?
Cyclic peptides found in natural peptide hormones We can find several cyclic peptides from natural peptide hormones such as calcitonin, oxytocin, somatostatin, vasopressin, and so on. These peptides form rigid structure by forming disulfide bond connecting two Cys residues in the peptide.
What are peptides explain solid-phase Peptide Synthesis?