Fats and fatty acids are polymerized by oxidative or thermal processes. Structures have been deduced by using a number of chemical and physical techniques. In addition, urea fractionation and a number of chromatographic techniques are useful for the detection of monomers, dimers, and polymers.
What polymer is formed from fatty acids?
During the ester bond formation, three molecules are released. Since fats consist of three fatty acids and a glycerol, they are also called triacylglycerols or triglycerides. Triacylglycerols: Triacylglycerol is formed by the joining of three fatty acids to a glycerol backbone in a dehydration reaction.
Do saturated fats polymerize?
Note that a saturated fat like coconut oil, lard, or ghee will not work very well or at all for this, since it does not have any double bonds to undergo polymerization.
What are glycerol and fatty acids?
Glycerol is a small organic molecule with three hydroxyl (OH) groups, while a fatty acid consists of a long hydrocarbon chain attached to a carboxyl group. A typical fatty acid contains 12–18 carbons, though some may have as few as 4 or as many as 36.
What is the polymer of fatty acids and glycerol?
A lipid is a polymer composed of three fatty acid molecules and one glycerol molecule. Lipids produce a special polymer form which is considered to be a key component of cell membranes and hormones.
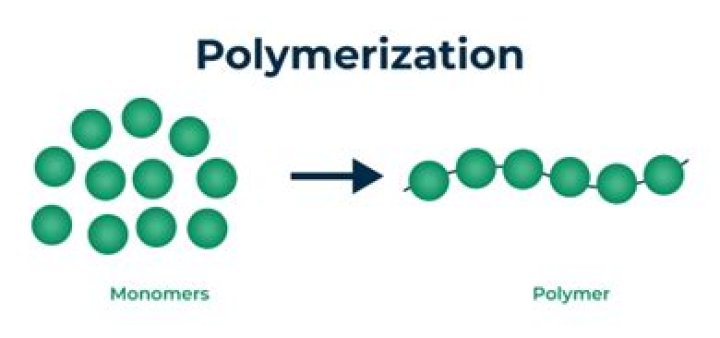
What is an example of polymerization?
Polymerization reactions are chain reactions, and the formation of Teflon from tetrafluoroethylene is one example. In this reaction, a peroxide (a compound in which two oxygen atoms are joined together by a single covalent bond) may be used as the initiator.
What is the important of polymerization?
A basic understanding of polymerization processes is important not only because polymerization affects structure, and hence properties, but also because some processing routes can convert monomers directly to a finished shape. They offer manufacturing industry considerable benefits both in direct and indirect costs.
What is the importance of polymerization?