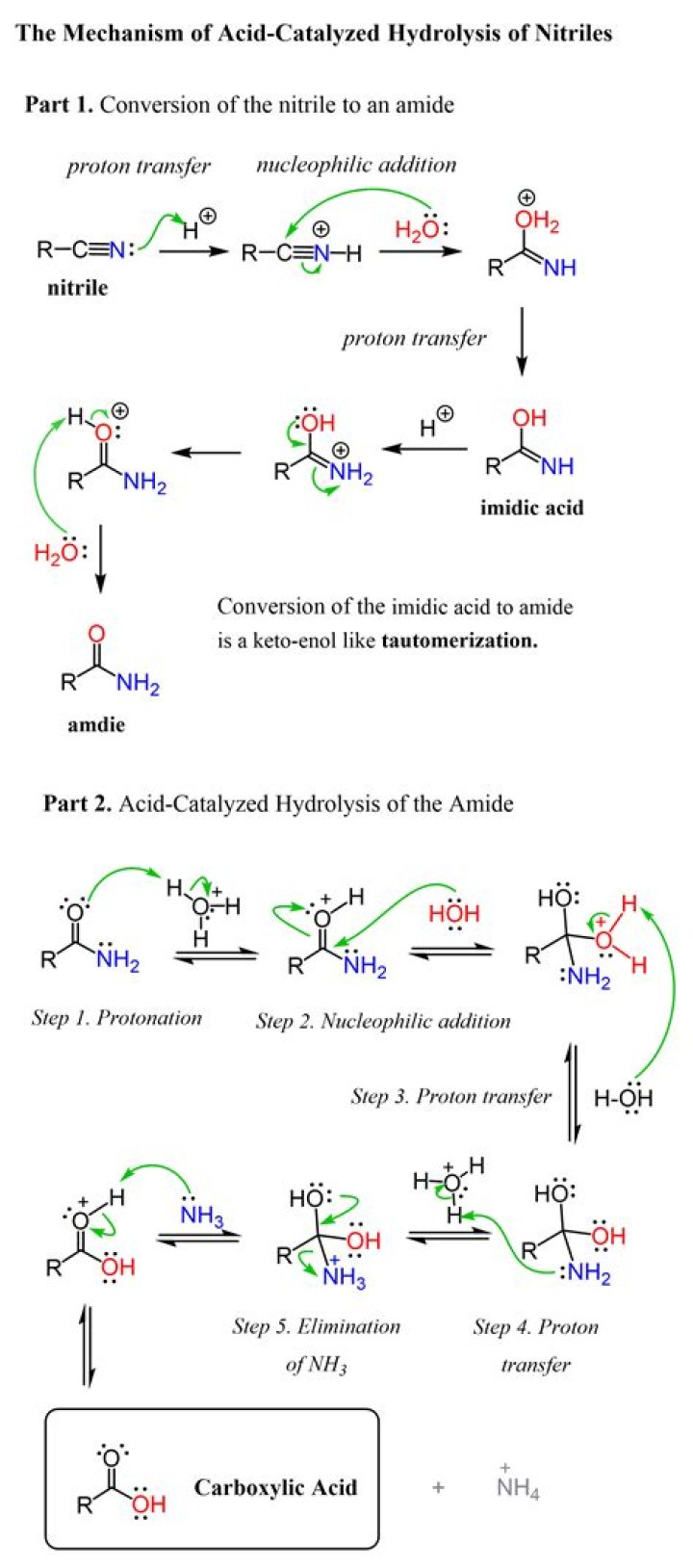
In chemistry, acid hydrolysis is a process in which a protic acid is used to catalyze the cleavage of a chemical bond via a nucleophile substitution reaction, with the addition of the elements of water (H2O). A common type of hydrolysis occurs when a salt of a weak acid or weak base (or both) is dissolved in water.
What are the products of acid hydrolysis?
However, acid hydrolysis process has been constantly explored to produce a variety of chemicals such as cellulose, hemicellulose, glucose, fructose, hydroxymethylfurfural, levulinic acid, xylose, and furfural [37,38].
What is acid and base hydrolysis?
Acidic hydrolysis is when water acts as a base to break apart a weak acid. Basic hydrolysis is when water acts as an acid to break apart a weak base. For example, water can react with urea, forming a hydroxide anion which can then release ammonia from the urea, which is critical for the growth of plants.
What are the three main types of hydrolysis?
2 There are three main types of hydrolysis: salt, acid, and base hydrolysis. Hydrolysis can also be thought of as the exact opposite reaction to condensation, which is the process whereby two molecules combine to form one larger molecule.
What are the two types of hydrolysis?
There are several types of hydrolysis, and we will look at them in brief below.
- Salts: This is the most common type of hydrolysis.
- Acid and Base: Acid–base-catalysed hydrolysis can be found during the hydrolysis of esters or amides.
What are the types of hydrolysis?
What is the mechanism of hydrolysis?
Usually hydrolysis is a chemical process in which a molecule of water is added to a substance. Sometimes this addition causes both substance and water molecule to split into two parts. In such reactions, one fragment of the target molecule (or parent molecule) gains a hydrogen ion.
What are the type of hydrolysis?
‘ There are three types of hydrolysis reactions: salt, acid, and base reactions. A salt hydrolysis involves the reaction between organic compounds and water. Acid and base hydrolysis involve the use of water as a catalyst to drive the hydrolysis reaction.
What enzyme is used in hydrolysis?
Enzymes that hydrolyze glycosidic bonds are called “glycoside hydrolases” or “glycosidases”. The best-known disaccharide is sucrose (table sugar). Hydrolysis of sucrose yields glucose and fructose. Invertase is a sucrase used industrially for the hydrolysis of sucrose to so-called invert sugar.
What is the role of acid in hydrolysis?
In organic chemistry, acid hydrolysis is a hydrolysis process in which a protic acid is used to catalyze the cleavage of a chemical bond via a nucleophilic substitution reaction, with the addition of the elements of water (H2O). For example, in the conversion of cellulose or starch to glucose. Formic acid. Nitric acid.
What is the process of hydrolysis?
In its simplest definition, hydrolysis is a chemical reaction in which water is used to break down the bonds of a particular substance. Hydrolysis can also be thought of as the exact opposite reaction to condensation, which is the process whereby two molecules combine to form one larger molecule.
Does hydrolysis remove or add water?
Hydrolysis can be the reverse of a condensation reaction in which two molecules join together into a larger one and eject a water molecule. Thus hydrolysis adds water to break down, whereas condensation builds up by removing water and any other solvents.
Is hydrolysis the same as digestion?
Many reactions of digestion are called hyrolysis reactions because they involve the reaction of a molecule of carbohydrate with a molecule of water, resulting in the carbohydrate, protein or fat molecule being broken into two new molecules. Hydrolysis is a Greek term that translates to “water separation.”.
What is required for a hydrolysis reaction?
Laboratory hydrolysis usually requires the use of a catalyst, which is typically a strong acid or alkali. Hydrolysis has been used for a long time in the production of soap . During this process, known as saponification, fat is hydrolyzed in a reaction with water and the strong alkali, sodium hydroxide.
What are the results of the hydrolysis reaction?
The process of hydrolysis, when applied to the chemical compound nitrile, will result in the creation of carboxylic acid and ammonia. This reaction happens in two stages. Hydrolysis can result in the synthesis of helpful chemicals. Sulfonylureas and organophosphate esters can be hydrolyzed into specific types of insect killers and pesticides.