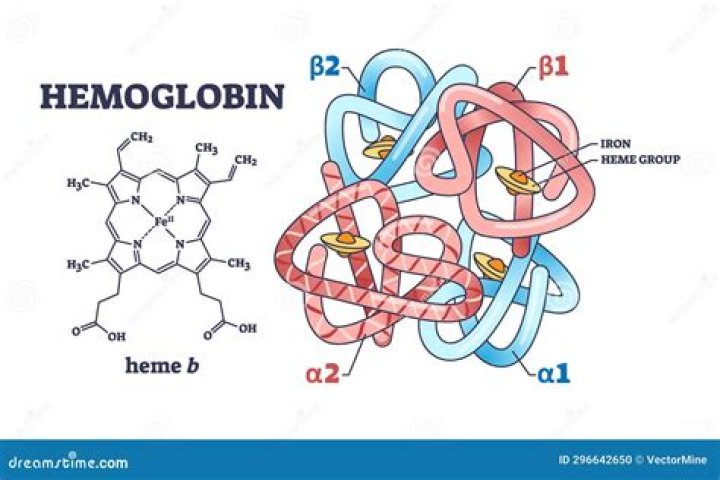
Heme iron is the type of iron found in blood and meat. Present in animal foods like red meats, fish, and poultry, heme iron is a significant source of the absorbed iron in a typical western diet, though it only makes up about one-third of dietary iron.
Is heme good for your health?
High heme intake is associated with increased risk of several cancers, including colorectal cancer, pancreatic cancer and lung cancer. Likewise, the evidence for increased risks of type-2 diabetes and coronary heart disease associated with high heme intake is compelling.
What is heme used for?
Hemes are used for two known reasons: to carry oxygen and to transport or store electrons. In the above image, you can see how gaseous oxygen can reversibly bind to the heme complex. Organisms use the heme molecule, in complex with specially-shaped proteins, to transport oxygen and move electrons.
How many mg of iron do you need a day?
The average daily iron intake from foods and supplements is 13.7–15.1 mg/day in children aged 2–11 years, 16.3 mg/day in children and teens aged 12–19 years, and 19.3–20.5 mg/day in men and 17.0–18.9 mg/day in women older than 19. The median dietary iron intake in pregnant women is 14.7 mg/day [5].
Is iron better absorbed with vitamin C?
Foods Rich in Vitamin C Vitamin C has been shown to enhance iron absorption. It captures non-heme iron and stores it in a form that’s more easily absorbed by your body ( 3 ). Foods high in vitamin C include citrus fruits, dark green leafy vegetables, bell peppers, melons and strawberries.
Why is heme iron bad?
Observational studies suggest that a high intake of heme iron may increase the risk of colon cancer ( 15 , 16 ). Clinical trials in humans have shown that heme iron from supplements or red meat may increase the formation of cancer-causing N-nitroso compounds in the digestive tract ( 17 , 18 ).
Is heme iron ferric or ferrous?
Dietary iron occurs in three major forms: ferrous iron (Fe2+), ferric iron (Fe3+), and heme iron (Fe2+ chelated into a complex organic compound to complete the heme structure, which occurs in hemoglobin, myoglobin, and certain enzymes).
Is heme a hemoglobin?
Heme is an iron-containing molecule that is important for many biological processes. Heme combines with globin proteins to form hemoglobin, which carries oxygen in red blood cells from the lungs to the rest of the body.
Why do cells need heme?
Heme serves as the prosthetic group of numerous hemoproteins (eg, hemoglobin, myoglobin, cytochromes, guanylate cyclase, and nitric oxide synthase) and plays an important role in controlling protein synthesis and cell differentiation.