Electrodialysis is a process by which electrically charged membranes. are used to separate ions from an aqueous solution Sy the driving force. of an electrical potential difference. Electrodialysis is used today mainly. for desalination of sea and brackish water.
What principle is used in electrodialysis process?
1. What principle is used in electrodialysis? Explanation: An electric field and ion selective membranes is used for separating an electrolyte feed in an dilute and concentrated desalted water. 2.
What is the purpose of electrodialysis?
Electrodialysis (ED) is used to transport salt ions from one solution through ion-exchange membranes to another solution under the influence of an applied electric current. It is an alternative to reverse osmosis (RO) as a desalinization mechanism.
What is electrodialysis water purification?
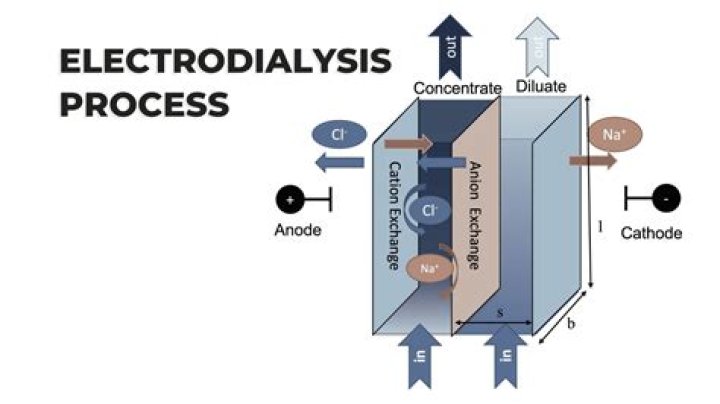
Electrodialysis is an electrochemical process in which ion transfer separates salt from water. In electrodialysis, filters or membranes selectively impervious to cations or anions are placed alternately between the electrodes (Figure 35).
What is electrodialysis colloidal solution?
Electrodialysis is the method for the purification of colloidal sol containing excess amount of electrolytes. From semipermeable membrane, a bag is made. It is filled with colloidal solution and suspended in fresh water. The electrolyte particles pass through the membrane and the colloidal solution is left behind.
How is RO different from electrodialysis?
Whereas RO is dependent on high pressure for its membrane treatment, ED works by cross-flow separation using ion exchange (IX) membranes, which is a low-pressure/tangential flow process.
How are the colloidal solution purified by electrodialysis?
Electrodialysis-. In the process of electrodialysis, The dialysis is carried out by under the influence of electric fields, to increase the process of purification. Ultra – filtration:- As pores are larger than the size of sol particles, Sol particles directly pass through ordinary filter paper.
How many cells are in a stack of water desalination systems?
In the video, what you see is actually a stack of two “cell pairs,” each consisting of a channel for the water that is being desalinated, and a channel where the brine is being concentrated. The number of cell pairs in an actual stack varies depending on the electrodialysis system, with as many as 600 cell pairs in a typical industry-scale system.
How do you design a desalination process using the simulation?
In this challenge, you will design a desalination process using the simulation. You will need to decide the dimensions of your cells and cell stack, the flow pressure level, and the amount of voltage to apply.
How does electrodialysis remove salt from water?
Salt in water takes the form of ions which, because they are charged, move under the influence of an electric field. Electrodialysis uses an applied electric field to remove these salt ions through ion-permeable membranes.
How does electelectrodialysis work?
Electrodialysis uses an applied electric field to remove these salt ions through ion-permeable membranes. The positive ions (cations) move in one direction while the negative ions (anions) move in the opposite direction.