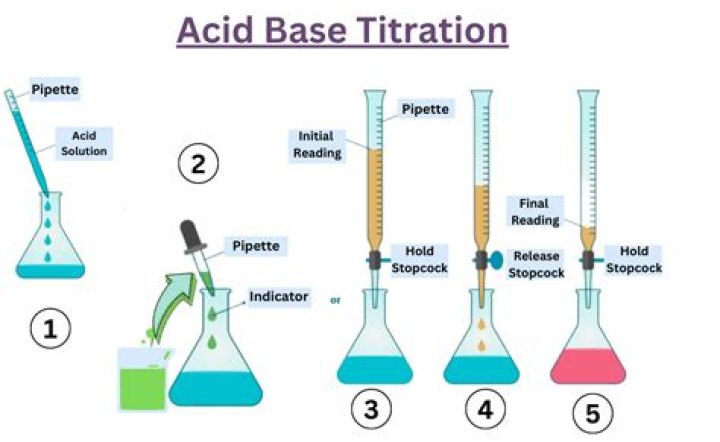
Potentiometric titration is a technique similar to direct titration of a redox reaction. It is a useful means of characterizing an acid. No indicator is used; instead the potential is measured across the analyte, typically an electrolyte solution.
What happens to the pH during an acid base titration?
Before any base is added, the pH of the acetic acid solution is greater than the pH of the [Math Processing Error] solution, and the pH changes more rapidly during the first part of the titration.
How do you calculate pH before titration?
If that number is greater than the number of moles of base B, the titration is past the equivalence point. To find the pH, first simply find the moles of excess H3O+. The excess can be calculated by subtracting initial moles of analyte B from moles of acidic titrant added, assuming a one-to-one stoichiometric ratio.
How do you calculate pH from titration?
produces a small amount of H3O+, the excess strong acid HCl dominates and determines the pH. If total H3O+ from HCl was 0.150 moles, then excess H3O+ = 0.050 moles. pH = [H3O+] (excess) pH = -log[H3O+] Excess strong acid results in pH < 7 (acidic).
What is the principle of potentiometric titration?
Potentiometric Titration principle of Potentiometry Principle. When the pair of electrodes are placed in the sample solution or analyte it shows the potential difference between two electrodes by addition of the titrant or by the change in the concentration of ions.
How does pH affect titration?
If you’re doing acid-base titration, pH plays a big role. It is usually done to determine the concentration of a solution or the molarity. pH indicates the end point. If you’re using titration to prepare a soluble salt, it may affect purity of the product obtained.
How does pH change during titration?
If we consider the titration of an acid by a base, the pH will increase. However, if we titrate a base by an acid, the pH will decrease. Example: The initial pH of the acidic solution HCl(aq) is around 1 .
How do you do potentiometric titration?
Potentiometric Titration is done via the usage of two electrodes – an indicator electrode and a reference electrode (generally a hydrogen electrode or a silver chloride electrode). One half-cell is formed with the indicator electrode and the ions of the analyte, which is generally an electrolyte solution.
When a pH probe is used during a titration?
A pH meter is used to measure the pH as base is added in small increments (called aliquots) to an acid solution. A graph is then made with pH along the vertical axis and volume of base added along the horizontal axis. From this graph the equivalence point can be determined and the molarity of the base calculated.
What is potentiometric acid-base titration?
Potentiometric Acid-Base Titration. Determination of Sodium Carbonate A hydroc hloric acid solution is standardized using primary standard sodium carbonate. A soda ash sample is then analyzed to determine its % Na 2CO 3. The end point is determined using a pH meter.
What is the purpose of a pH meter in titration?
In such cases, a pH meter can be used to monitor the acidity of the solution throughout the titration. The pH Meter (see Tro, p. 806) A pH meter consists of two electrodes: a glass electrode, which is sensitive to the concentration of hydronium ions in solution, and a reference electrode.
What are the different types of titration?
There are four types of titration that fall under the category of potentiometric titration, namely acid-base titration, redox titration, complexometric titration, and precipitation titration. A brief description of each of these types of titration is given below.
What is the use of precipitate titration?
This method is used to determine a mixture of metal ions in a given solution. Precipitation Titration: This type of titration involves a reaction between the given analyte and the titrant wherein an insoluble precipitate is formed. The end-point of this titration is noted when the addition of the titrant no longer forms a precipitate.