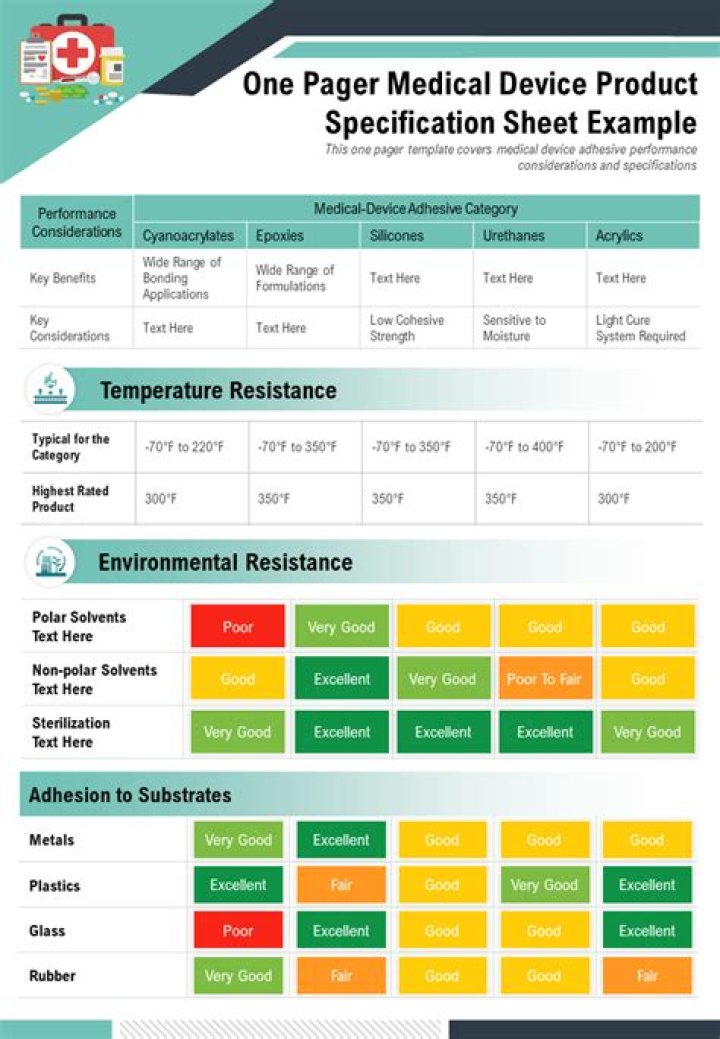
A product requirement specification outlines what needs to be achieved during the medical device design process. It is an important step in the process as it ensures design engineers fully understand what is required so they can deliver the best possible product.
What are standards in medical devices?
Some of the most common reference standards for device manufacturers to follow include: ISO 13485, which is the standard for medical device quality management systems. ISO 13485:2016 is the most current version, and it’s also harmonized in Europe. ISO 14971, which is the standard for medical device risk management.
How do you write a medical device requirement?
When writing the PRD and System requirements, each requirement should be testable and measurable. For example, rather than having a requirement “The system will inflate a balloon.”, a measurable requirement would be: The flow rate from the system will inflate the balloon to 4 psi in four seconds.
Why are FDA design guidelines so non prescriptive when relating to medical devices?
Because design controls must apply to a wide variety of devices, the regulation does not prescribe the practices that must be used. Instead, it establishes a framework that manufacturers must use when developing and implementing design controls.
What makes PRD good?
PRDs typically include the following elements: Purpose — Who it’s for and why you’re building it. Features — What you’re going to build. Release Criteria — Goals for the release (e.g., functionality).
What is the standard used in medical devices industry?
ISO 13485
ISO 13485 is designed to be used by organizations involved in the design, production, installation and servicing of medical devices and related services. It can also be used by internal and external parties, such as certification bodies, to help them with their auditing processes.
How do you design a medical device?
Design Stages. Medical device design typically goes through six stages. These are research and discovery, specification development, engineering, prototyping, iteration, and manufacturing process design.
What is a medical device file?
Medical device files are documents that includes descriptions of design records, manufacturing processes, product specifications, device usage guides, quality measurement criteria, levels of compliance with regulatory bodies and quality standards, and, if required, servicing and installation records and their …
What are the main essentials for writing the requirements?
Table of Contents
- Ensure each requirement is specific.
- Define requirements in measurable terms.
- Use imperatives such as “shall” or “must” properly and consistently.
- Enforce consistency of terminology and prohibit industry or company jargon.
- Prohibit passive voice.
- Avoid duplicate or contradictory requirements.
What is design output medical device?
A Design Output is a drawing or specification or manufacturing instruction. Design Outputs describe all the components, parts, and pieces that go into your medical device. Design Outputs describe all assemblies and sub-assemblies of your product.
Which law placed medical devices under FDA oversight for the first time?
the Medical Device Amendments Act
FDA was given the authority to begin regulating all medical devices on May 28, 1976. This is when the President signed the Medical Device Amendments Act.
How do you write a medical device essential requirements checklist?
When writing a medical device essential requirements checklist, it is important to keep in mind that you must be able to demonstrate how the requirement is met. If you cannot quickly come up with an objective way to show that the requirement has been met, it probably needs to be rewritten.
What are Medical Device Standards and regulations?
In order to get into the market, the medical device needs to pass through certain regulatory compliances, subject to both regional and international standards. Medical device standards are helpful and enforced by law in specifying and evaluating the requirement for design and performance parameters for a biomedical materials, tools, and equipment.
What are the criteria for medical device classification?
Medical device classification is based on the risk associated with the use and enforced by law. Also, you need to look for any pre-existing Intellectual Property on the proposed idea or similar; it might actually disallow you to use the mechanism or technology.
What are the steps in the medical device design process?
This phase consists of initial designing, prototyping, PoCs, and iteration driven redesign. Once you successfully complete the product conceptualization and discovery phase, you may proceed further for FDA approval and commercialization. What are some Biggest Challenges in Medical Device Design?