Now consider what happens if NaOH functions as a base. It will remove the most acidic H in the system which is the one in the -OH group. This creates an alkoxide which is a Nu and can displace the Br intramolecularly to give an epoxide. The stereochemistry in dictated by the fact that this is an SN2 type process.
How do you break up epoxides?
Epoxides can also be opened by other anhydrous acids (HX) to form a trans halohydrin. When both the epoxide carbons are either primary or secondary the halogen anion will attack the less substituted carbon and an S N2 like reaction.
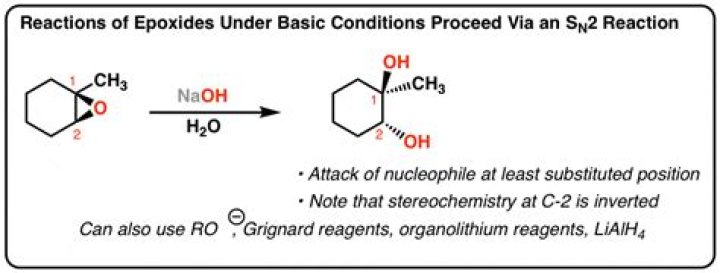
How do nucleophiles react with epoxides also known as Oxiranes under basic conditions?
Reactive nucleophiles react with epoxides in an SN2 type of reaction. Reactive nucleophiles are usually anions so the reaction conditions are essentially basic. The leaving group is the oxygen atom of the epoxide in the form of the alkoxide which is converted to the alcohol on an acidic work-up.
Which reagent can convert epoxide ring into alcohol?
The reaction of epoxides with TMSCN (an organic-soluble and easier-to-handle substitute to traditional cyanide reagents such as KCN) and catalytic ZnI2 gives 1,2-isocyano alcohols.
What reactions do ethers undergo?
Ethers are fairly unreactive, but they do react in three ways:
- Chlorination – an aromatic ether reacts with chlorine to form 2,4,6-trichloroanisole.
- Oxidation – an ether will slowly react with oxygen in the air to form explosive peroxides, molecules with two oxygen atoms attached to each other.
Why can epoxides react with Grignards?
Due to the three-membered-ring of an epoxide, a lot of energy is ‘stored’ in ring strain. This destabilizes the stucture and is a driving force for a Grignard reaction (or any substitution on an epoxide). The THF is a five-membered-ring, which is far more stable.
What reagent breaks an epoxide?
Epoxides can also be opened by other anhydrous acids (HX) to form a trans halohydrin. When both the epoxide carbons are either primary or secondary the halogen anion will attack the less substituted carbon and an SN2 like reaction.