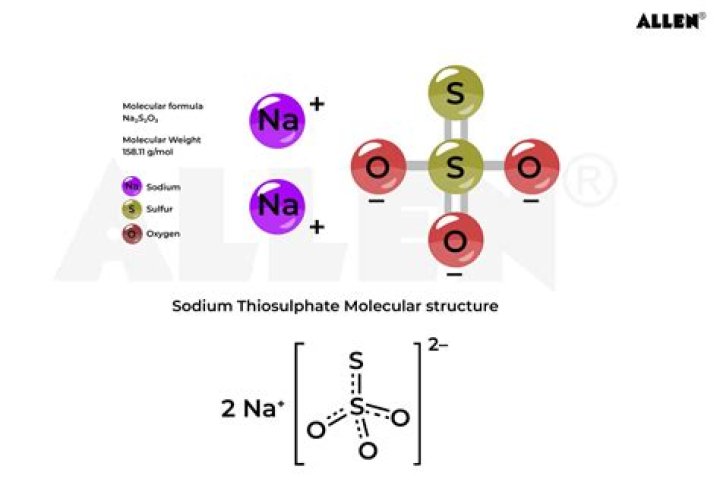
Explanation: To calculate the molar mass of sodium thiosulfate, Na2S2O3 , we need to find all the molar masses of the elements, and add them together. Na has a molar mass of 23 g/mol . So, Na2 will have a molar mass of 23 g/mol⋅2=46 g/mol .
How do you find the molarity of Na2S2O3?
- solution of sodium thiosulfate of volume V=50ml and molar concentration C=0.1mol/l is prepared by dissolving a mass(m) of solid penta-hydrated sodium thiosulfate Na2S2.
- Solution:
- Firstly, we calculate the molar mass of Na2S2O3 and Na2S2O3.
- .5H2O:
- M(Na2S2O3)= 23·2+32·2+16·3=158 g/mol;
- .
How do you find the molar mass of thiosulfate?
112.1282 g/mol
Thiosulfate/Molar mass
What is molar mass of sodium?
22.989769 u
Sodium/Atomic mass
How do you find the equivalent mass of Na2S2O3?
What is the equivalent weight of sodium thiosulphate? The weight equivalent is the gram molecular weight divided by the number of electrons lost or obtained by each molecule; this is (248.17/1) g for sodium thiosulfate (Na2S2O3.
What is the molar mass of sodium bicarbonate nahco3?
84.007 g/mol
Sodium bicarbonate/Molar mass
What is the molar mass of 2nacl?
The molar mass of sodium chloride is 58.44 g/mol.
What’s the molar mass of sodium bromide?
102.894 g/mol
Sodium bromide/Molar mass
What is the equivalent mass of sodium?
Instead, these chemists had settled on a list of what were universally called “equivalents” (H = 1, O = 8, C = 6, S = 16, Cl = 35.5, Na = 23, Ca = 20, and so on).
What is the equivalent number of Na2S2O3?
248
Therefore, the equivalent weight of sodium thiosulphate is 248.
What is the formula mass for Na2SO3?
1) Na2SO3 = 2Na(+) + SO3 (2-) => 1 mol SO3 (2-) : 1 mol Na2SO3. 2) Molar mass of Na2 SO3. 2*23 g/mol + 32g/mol + 3*16 g/mol = 126 g/mol. 3) moles of Na2SO3. n = grams / molar mass = 99.6mg * [1 g /1000 mg] / 126 g / mol = 7.9*10^-4 mol Na2SOs.
How do you calculate the molar mass of a gas?
The molar mass of a particular gas is therefore equal to the mass of a single particle of that gas multiplied by Avogadro ’s number (6.02 x 1023 ). To find the molar mass of a mixture of gases, you need to take into account the molar mass of each gas in the mixture, as well as their relative proportion.
How do you calculate molar mass in chemistry?
The molar mass is the mass of one mole of a sample. To find the molar mass, add the atomic masses (atomic weights) of all of the atoms in the molecule. Find the atomic mass for each element by using the mass given in the Periodic Table or table of atomic weights.
Are molar mass and molecular mass the same thing?
Molar mass is the same as molecular weight. However, the main difference is that molecular weight only describes the weight of the molecules over the elements. First, molar mass is defined as the mass of a mole or one mole.