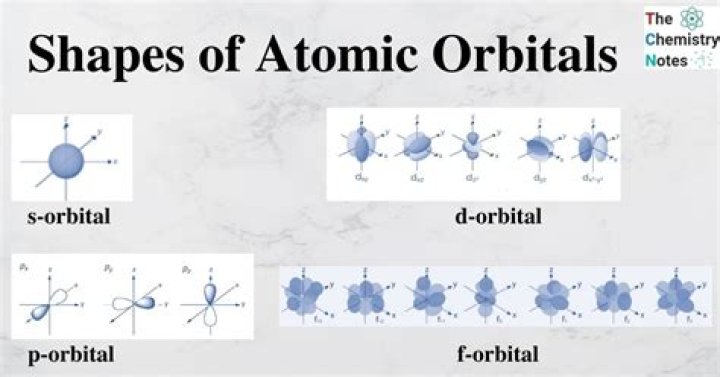
Named for their energy sublevels, there are four types of orbitals: s, p, d, and f. Each orbital type has a unique shape based on the energy of its electrons. The s orbital is a spherical shape. The p orbital is a dumbbell shape.
What are the shapes of atomic orbitals?
An s-orbital is spherical with the nucleus at its centre, a p-orbitals is dumbbell-shaped and four of the five d orbitals are cloverleaf shaped. The fifth d orbital is shaped like an elongated dumbbell with a doughnut around its middle. The orbitals in an atom are organized into different layers or electron shells.
How do you determine the shape of an orbital?
The angular quantum number (l) describes the shape of the orbital. Orbitals have shapes that are best described as spherical (l = 0), polar (l = 1), or cloverleaf (l = 2). They can even take on more complex shapes as the value of the angular quantum number becomes larger.
What does this orbital shape represent?
The orbital shape represents the region in space where the electron is most likely to be found. The different lobes of an orbital are separated by regions in space where the probability of finding an electron is zero.
What is an atomic orbital in chemistry class 11?
Atomic orbitals are the 3-dimensional spaces around the nucleus where the probability of finding an electron is maximum. The molecules orbitals are formed by combining the atomic orbitals. In quantum chemistry we have encounter orbitals which are s, p, d and f subshells.
What is an atomic orbital in chemistry?
orbital, in chemistry and physics, a mathematical expression, called a wave function, that describes properties characteristic of no more than two electrons in the vicinity of an atomic nucleus or of a system of nuclei as in a molecule. An s orbital is spherical with its centre at the nucleus.
How does an atomic orbital differ from an orbit?
Differences between Orbit and Orbitals An orbit is the simple planar representation of an electron. An orbital refers to the dimensional motion of an electron around the nucleus in a three-dimensional motion. An orbital can simply be defined as the space or the region where the electron is likely to be found the most.
What are some examples of atomic orbitals?
For example, if the value of ‘n’ is equal to 3, the possible values of ‘l’, which range from zero to (3-1), are 0, 1, and 2. The names of these atomic orbitals will be 3s (for n=3 and l=0), 3p (for n=3 and l=1), and 3d (for n=3 and l=2).
What are atomic orbital examples?
Orbital Definition and Example Orbital Definition. In chemistry and quantum mechanics, an orbital is a mathematical function that describes the wave-like behavior of an electron, electron pair, or (less commonly) nucleons. Orbital Examples. The 1s 2 orbital contains two electrons. Properties of Electrons in Orbitals. Orbitals and the Atomic Nucleus.
What are the four orbitals called in an atom?
The s orbital, where the value of the azimuthal quantum number is equal to 0. The p orbital, where the value of the azimuthal quantum number is equal to 1. The d orbital, where the value of the azimuthal quantum number is equal to 2. The f orbital, where the value of the azimuthal quantum number is equal to 3.
How many orbitals are in an atom?
For any atom, there are seven 6f orbitals. The f-orbitals are unusual in that there are two sets of orbitals in common use. Those shown here are the cubic set and these are appropriate to use if the atom is in a cubic environment, for instance.