T Toxic by ingestion and inhalation with a danger of cumulative effects. Liberates highly toxic hydrogen selenide in contact with gastric juices. Dermatitis may result from prolonged contact. Particular care must be exercised when machining and creating dust or particles.
What is zinc selenide used for?
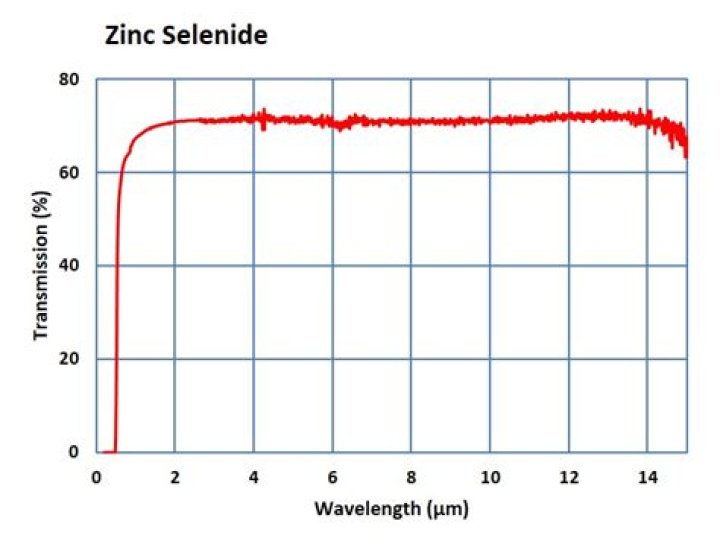
Zinc Selenide is used for infrared windows, lenses, and prisms where transmission in the range 0.63µm to 18µm is desired. Zinc Selenide has a very low absorption co-efficient and is used extensively for high power infrared laser optics. It is non-hygroscopic.
Is zinc selenide a semiconductor?
Zinc selenide (ZnSe), is a light yellow binary solid compound. It is an intrinsic semiconductor with a band gap of about 2.82 eV at 25 °C. ZnSe rarely occurs in nature.
What is the physical state of zinc selenide?
solid
Zinc selenide (ZnSe) is a light-yellow, solid compound comprising zinc (Zn) and selenium (Se)….Zinc selenide.
| Names | |
|---|---|
| Appearance | light yellow solid |
| Density | 5.27 g/cm3 |
| Melting point | 1,525 °C (2,777 °F) |
| Solubility in water | negligible |
How is zinc selenide made?
Zinc Selenide is produced by synthesis from Zinc vapour and H2Se gas, forming as sheets on Graphite susceptors. Zinc Selenide oxidizes significantly at 300°C, exhibits plastic deformation at about 500°C and dissociates about 700°C. For safety, Zinc Selenide windows should not be used above 250°C in normal atmosphere.
What is zinc and selenium good for?
Selenium and zinc are essential micronutrients that have important roles in reducing oxidative stress and protecting DNA from the attack of reactive oxygen species. Selenium is an essential trace element that possesses several functions in many cellular processes for cancer prevention.
Is zinc a sulfide?
Zinc sulfide (or zinc sulphide) is an inorganic compound with the chemical formula of ZnS. This is the main form of zinc found in nature, where it mainly occurs as the mineral sphalerite. In its dense synthetic form, zinc sulfide can be transparent, and it is used as a window for visible optics and infrared optics.
Is zinc selenide ionic?
There are two extreme ways of thinking of ZnSe. One is as a purely ionic system, with Zn2+ and Se2- ions.
Is it OK to take zinc and selenium together?
No interactions were found between selenium and Zinc.
Does selenium interfere with zinc absorption?
Selenium had an antagonistic effect on zinc absorption by zinc-depleted rats, and zinc had an antagonistic effect on selenium absorption by zinc-adequate rats.
Is zinc sulfide safe?
Slightly toxic by ingestion. Target organs: None known. This material is considered hazardous by the OSHA Hazard Communication Standard (29 CFR 1910.1200). Zinc Sulfide (1314-98-3), 98-100% .
Can zinc selenide be used in harsh environments?
Since Zinc Selenide is a relatively soft material that scratches and digs can be easily remained on the surfaces during processing flow, it is not recommended for use within harsh environments, Hyperion Optics’ advanced manufacturing techniques for ZnSe ensure superior surface quality compared to our competitors.
What are the optical properties of zinc selenide?
Zinc Selenide (ZnSe) Optical Properties Bulk Absorption Coefficient @ 10.6 µm ≤ 0.0005 cm -1 Temperature Change of Refractive Index @ 61 x 10 -6 /°C Refractive Index Inhomogeneity @ 632.8 n < 3 x 10 -6 Refractive Index Values ± 0.001
Are ZnSe lenses available with AR coating?
ZnSe aspheric lenses are also available for your application; please refer toIR Aspheric Lenses for more information. ZnSe Dome optics are also available in ourDomecategory. Our ZnSe lenses are also available with AR coating according to specific requirement.
Is ZnSe safe to use?
It’s safe to use in most industrial, field, and laboratory environments. II-VI has the capability to grow prism grade ZnSe up to 2.50” thick. Prism grade ZnSe exhibits minimal refractive index variations within the material on planes perpendicular to the growth direction as well as in other directions.