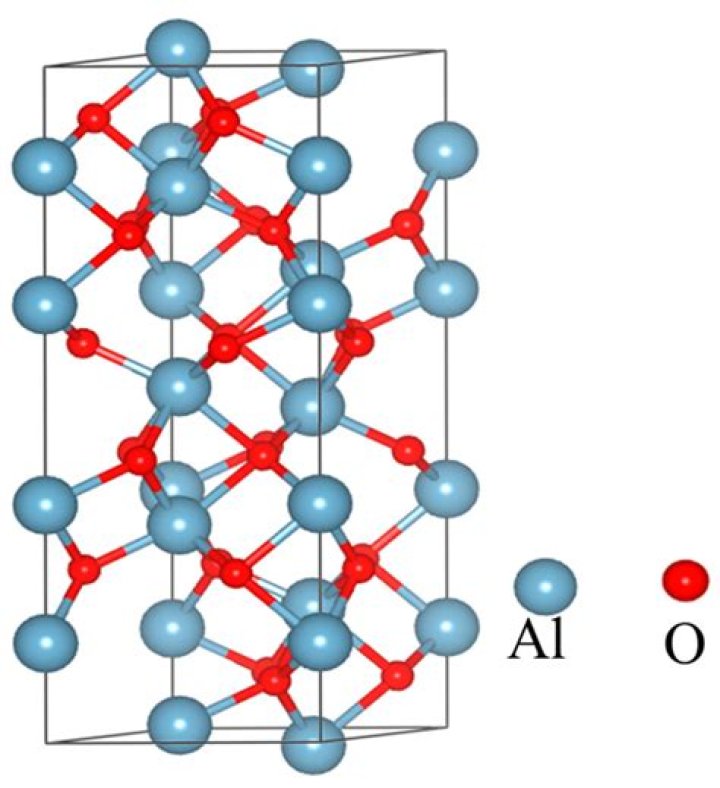
nglos324 – al2o3. Aluminum oxide is a ceramic compoundwith a hexagonal crystal lattice. Because of the high charge (3+) on the cations, the aluminum ions want to have the maximum spacing possible in the structure.
What are involved in Born Haber cycle?
Born Haber process or more commonly referred to as Born Haber cycle is a method that allows us to observe and analyze energies in a reaction. It also involves several steps or processes such as electron affinity, ionization energy, sublimation energy, the heat of formation and dissociation energy.
Why does Al2O3 have high lattice energy?
Al2O3 has the higher lattice energy than that of Ga2O3 , because Al3+ is a smaller cation than Ga3+ (one less quantum level, so the electrons are attracted in by more). Na2O has the higher lattice energy than NaOH because the charge of O2− is larger than that of OH− . All other factors are the same.
What type of bond is Al2O3?
Al2O3 is ionic due to relative size of oxygen and aluminium and polarizing power of Al, (since we know that aluminium has a charge of +3, provides three electrons) in case of Al2Cl6 & AlCl3 , it seems to be covalent due to similarities like banana bonding & bigger radius of Cl (in compression to oxygen).
How is Al2O3 formed?
In terms of electron arrangement in the formation of the ionic compound aluminium oxide, two aluminium atoms donate their three outer electrons to three oxygen atoms. This results in two triple positive aluminium ions to three double negative oxide ions via electron transfer.
What is born Haber cycle How does it explain the stability of ionic compounds?
Ionic solids tend to be very stable compounds. The enthalpies of formation of the ionic molecules cannot alone account for this stability. These compounds have an additional stability due to the lattice energy of the solid structure.
Which step in the Born Haber cycle will typically be endothermic?
As you can see in the accompanying diagram, the first step involves formation of gaseous sodium metal from solid sodium metal. This is called the sublimation energy of sodium, and represented by Hsub. This step is endothermic, with sublimation energy of sodium being +108 KJ.
What is born Haber cycle explain with example?
Describe the Born – Haber cycle This will be used as the energy of ionic solids in the equation. Step 2: According to the Born–Haber cycle, the elements shall be in the gaseous forms. Add the enthalpy change by turning the elements into the gaseous states.
Does Al2O3 have high lattice energy?
Lattice energy is the enthalpy change that occurs when 1 mol of ionic solid separates into gaseous ions. The hardness of Al2O3 implies that there is a strong interaction between its ions and that high amount of energy will be required to separate them. Hence, Al2O3 has a high lattice energy.
Why MgO is more stable than Al2O3?
The Gibbs energy of formation of MgO is most negative and the line for oxidation of Mg lies lower than the line for oxidation of Al, which means Mg can reduce Al2O3 to Al. Hence MgO is more stable than Al2O3.
Is Al2O3 polar covalent bond?
Talking with respect to polarity, Al2O3 definitely possesses polar behaviour because of the difference in the electronegativity of the two constituting elements and because of the ionic nature.