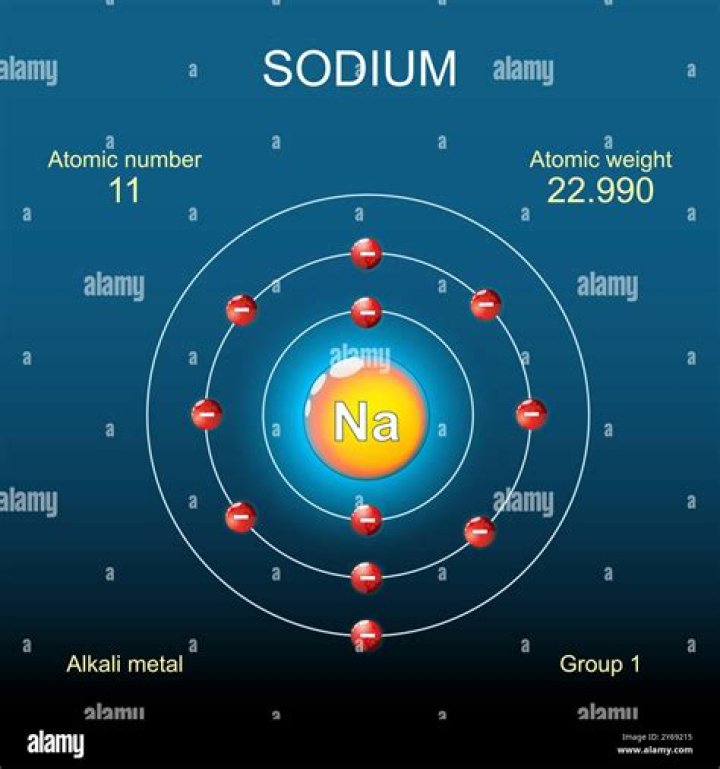
When we write the configuration we’ll put all 11 electrons in orbitals around the nucleus of the Sodium atom. In writing the electron configuration for sodium the first two electrons will go in the 1s orbital. Since 1s can only hold two electrons the next 2 electrons for sodium go in the 2s orbital.
What is the correct electron configuration for sodium NA )?
[Ne] 3s¹
Sodium/Electron configuration
What is the atom of sodium chloride?
Chemically, table salt consists of two elements, sodium (Na) and chloride (Cl). Neither element occurs separately and free in nature, but are found bound together as the compound sodium chloride.
What is the orbital diagram for sodium?
The orbital diagram of the sodium atom is shown below. The sodium atom has 11 electrons which are contained in 1s, 2s, 2p and 3s orbitals.
What is the formation of sodium chloride?
Sodium chloride is formed when sodium atoms interact with chlorine atoms. Sodium will donate an electron (which is a negatively-charged particle) to chlorine. This makes sodium slightly positive and chlorine slightly negative.
How many orbitals does chlorine have?
In it, there’s only 1 orbital. Since 1 orbital can hold at most 2 electrons, therefore 1s can hold max 2 electrons . For energy level 2, there are 2 sublevels, 2s and 2p .
How is Na+ formed from a sodium atom?
Neutral sodium atom (Na) becomes sodium cation (Na+) by releasing an electron. positive charge on the sodium cation is balanced by the negative charge on the chloride, so the ionic compound is neutral.
What is the electron configuration of a sodium atom in CL17?
The sodium electron configuration is 1s 2 2s 2 2p 6 3s 1. The electron configuration of chlorine is Cl (17) = 1s 2 2s 2 2p 6 3s 2 3p 5. The electron configuration of a sodium atom indicates that there is one (3s 1) electron in the 3rd orbit of the sodium atom.
What is the electron configuration of the element na?
The symbol for sodium is ‘Na’. The electron configuration of sodium shows that the period of sodium is 3 and sodium is a s-block element. Sodium electron configuration with an orbital diagram is the main topic of this article. The eleventh element in the periodic table is sodium.
What is the number of electrons in sodium atom?
The atomic number of sodium is eleven and the total number of electrons in the sodium atom is 11. These electrons are arranged according to specific rules of different orbits. The position of the electrons in different energy levels of the atom and the orbital in a certain order is called electron configuration.
What is the sodium electron configuration in the Aufbau principle?
The sodium electron configuration in the Aufbau principle is 1s 2 2s 2 2p 6 3s 1. Another method of electron configuration is the Hund principle. The German physicist Friedrich Hund provided a guideline for the entry of electrons into different orbitals of equal power. Which is known as the Hund principle.