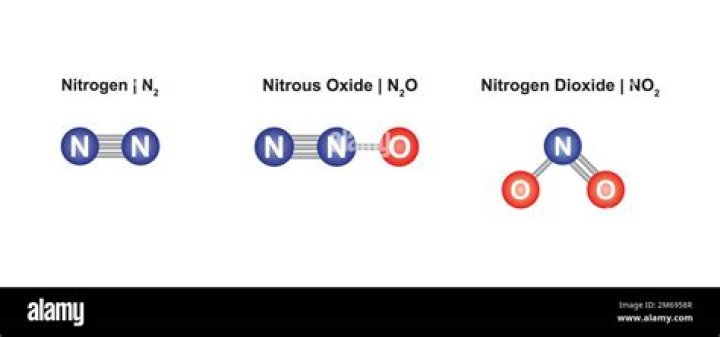
A substance’s molar mass tells you the mass of one mole of that substance. In your case, nitrogen dioxide, NO2 , is said to have a molar mass of 46.01 g/mol . That means that every mole of nitrogen dioxide will have a mass of 46.01 g .
What is the mass of one mole of nitrogen dioxide?
46.0055 g/mol
Nitrogen dioxide/Molar mass
How many moles is 84 g of nitrogen dioxide?
Therefore 6 moles of N atoms contain 84 grams …
How many particles are there in 1 mole of nitrogen?
In order to have one mole of things, let’s say particles, you need to have 6.022⋅1023 particles → this is known as Avogadro’s constant and acts as the definition of the mole. So, in one mole of nitrogen gas you have 6.022⋅1023 molecules of nitrogen gas, N2 .
How many moles are contained in 14 grams of nitrogen dioxide?
grams is the molar mass of nitrogen meaning that one mole of nitrogen atoms is 14 grams. One mole of any substance has 6.02*10^23 atoms/molecules of that subtance therefore there are 6.02*10^23 atoms of nitrogen in 14g of nitrogen.
What is the mass of 3 moles of nitrogen dioxide?
A certain gas takes five times as long to effuse outas H2(g) under identical conditions. Its molecular mass will be.
Is the molar mass of nitrogen dioxide?
What is the number of moles equal to?
One mole of a substance is equal to 6.022 × 10²³ units of that substance (such as atoms, molecules, or ions). The number 6.022 × 10²³ is known as Avogadro’s number or Avogadro’s constant.
How do you find the moles of N in n2o?
To establish the moles of N, we simply use the subscripts from the chemical formula of nitrous oxide. The chemical formula shows that 2 moles of N are present per mole of the compound.
What is the mole of co2?
44.01amu
The molecular mass of carbon dioxide is 44.01amu. The molar mass of any compound is the mass in grams of one mole of that compound. One mole of carbon dioxide molecules has a mass of 44.01g, while one mole of sodium sulfide formula units has a mass of 78.04g. The molar masses are 44.01g/mol and 78.04g/mol respectively.