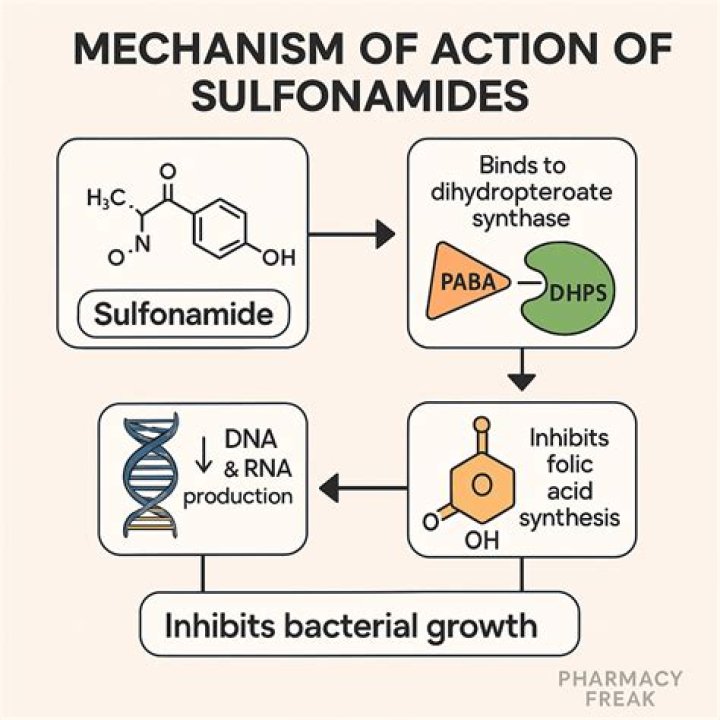
Sulfa Drug Synthesis The corresponding acetanilide undergoes chlorosulfonation. The resulting 4-acetamidobenzenesulfanyl chloride is treated with ammonia to replace the chlorine with an amino group and affords 4-acetamidobenzenesulfonamide. The subsequent hydrolysis of the sulfonamide produces the sulfanilamide.
Which catalyst is used for synthesis of sulfanilamide?
The reaction between sulfonyl halide and amines is usually catalyzed by basic catalyst such as sodium carbonate, potassium carbonate, pyridine or triethyl amine. The sulfonyl chloride method represents the most simple and direct route for the preparation of sulfonamides.
When acetanilide reacts with Chlorosulphonic acid what does it give?
This is the first step of the processes where required amount of acetanilide is reacted with chlorosulfonic acid to obtain an intermediate called p-acetamidobenzenesulfonyl chloride. This mixture is made to react in a reflux condenser for about 15-20 min where a semisolid paste is obtained which is brown.
Which is the first step in formation of sulfanilamide from Acetanilide?
Principle: Sulphanilamide can beprepared by taking acetanilide and treating it with excess of chlorosulphonic acid, which gives p-acetamidobenzenesulphonyl chloride which readily converted into corresponding p-acetamidobenzenesulphonamides upon reaction with ammonia or ammonium carbonate.
What is the chemical formula of sulfonamide?
Sulfanilamide | C6H8N2O2S – PubChem.
Which is the first step in formation of sulfanilamide from acetanilide?
What is the molar mass of acetanilide?
135.17 g/mol
Acetanilide/Molar mass
Which reagent is used for synthesis of sulfanilamide?
Sulphanilamide can beprepared by taking acetanilide and treating it with excess of chlorosulphonic acid, which gives p-acetamidobenzenesulphonyl chloride which readily converted into corresponding p-acetamidobenzenesulphonamides upon reaction with ammonia or ammonium carbonate.
What are the reagents used in the experiment of sulfanilamide?
Materials and Equipment – Reagents: Sulphanilamide solution. Dissolve 5g of NH2C6H4SO2NH2 (sulphanilamide) in 50 ml conc. HCl and about 300 ml DI. Dilute with DI to a final volume of 500 ml (a 1.2 N HCl solution can be purchased and substituted for conc.