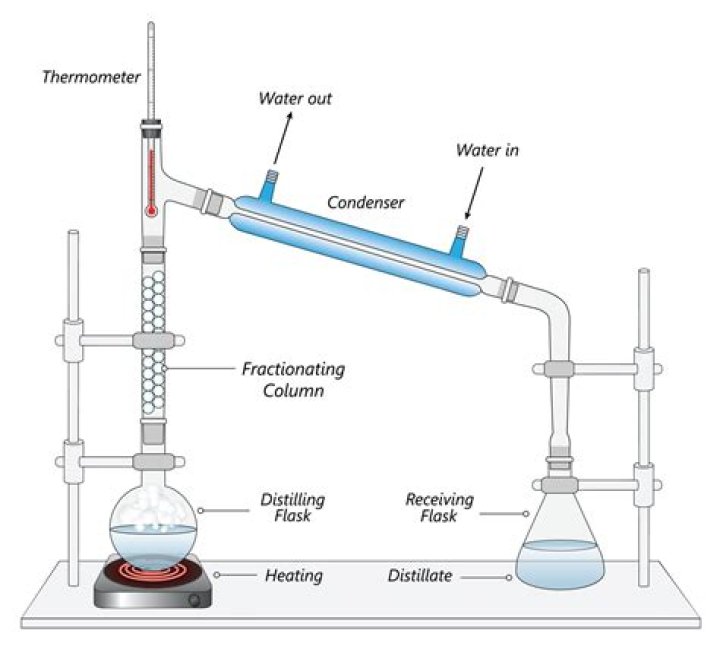
The liquid nitrogen and oxygen are then separated by fractional distillation . The liquefied air is passed into the bottom of a fractionating column. Just as in the columns used to separate oil fractions , the column is warmer at the bottom than it is at the top.
What is obtained from fractional distillation of air?
Fractional distillation of air. Many gases are obtained in commercial quantities through a process known as fractional distillation of liquefied air. Air is filtered to remove dust and other solids, water vapour and carbon dioxide. The gas mixture is then purified by fractional distillation.
How is nitrogen gas separated from air?
By a process called fractional distillation of liquid air to produce nitrogen. In simple terms, a four-step process is used: cool it, isolate the nitrogen, separate it from the air, and then collect it. Once in its liquid form, the gases can be transported in tanks, and stored in cylinders.
Is it possible to obtain oxygen from air by fractional distillation?
it is possible to obtain oxygen from air by fractional distillatio | Filo. Solution: Oxygen gas is prepared by fractional distillation of air. During this process, dinitrogen with less boiling point (78K) distills as vapours while dioxygen with higher boiling point 90 K) remains in the liquid state and can be separated …
Why oxygen can be separated from nitrogen by fractional distillation of liquid air?
The air in the Earth’s atmosphere consists of nitrogen, argon, oxygen, carbon dioxide and many other gases. So, oxygen can be separated from nitrogen by fractional distillation of liquid air because the two elements have different boiling points. Hence, option C is correct.
How is nitrogen obtained fractional distillation?
Nitrogen is produced commercially almost exclusively from air, most commonly by the fractional distillation of liquid air. In this process, air is first cooled to a temperature below that of the boiling points of its major components, a temperature somewhat less than – 328°F (-200°C).
Can oxygen be produced from nitrogen?
Conversion of Nitrogen Gas Plants to Produce Medical Grade Oxygen. existing nitrogen plants can be modified to produce oxygen. With the availability of ZMS, such modified plant can be set-up in 4-5 days while installation of new oxygen plant may take minimum 3-4 weeks.
Why oxygen can be separated from nitrogen by fractional distillation?
The air in the Earth’s atmosphere consists of nitrogen, argon, oxygen, carbon dioxide and many other gases. Some compounds such as carbon dioxide never liquefy. So, oxygen can be separated from nitrogen by fractional distillation of liquid air because the two elements have different boiling points.
Why is fractional distillation used to obtain oxygen from the air?
The most economical method for commercial preparation of O2 involves liquefaction of air (after removing CO2 and water vapour) followed by fractional distillation of the liquids air so obtained.
Can nitrogen be converted to oxygen?
It is possible to convert existing Pressure Swing Adsorption (PSA) based nitrogen plants to produce oxygen by replacing Carbon Molecular Sieve (CMS) used production of nitrogen with Zeolite Molecular Sieve (ZMS) and few other changes such as oxygen analysers, control panel system, flow valves etc.