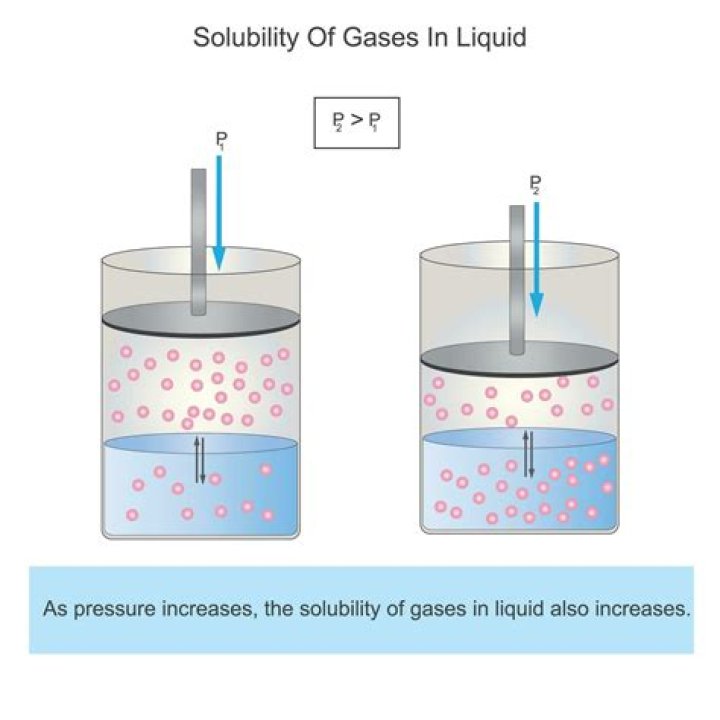
External pressure has very little effect on the solubility of liquids and solids. In contrast, the solubility of gases increases as the partial pressure of the gas above a solution increases.
How does pressure affect solubility of a gas in a liquid?
The solubility is a measure of the concentration of the dissolved gas particles in the liquid and is a function of the gas pressure. As you increase the pressure of a gas, the collision frequency increases and thus the solubility goes up, as you decrease the pressure, the solubility goes down..
Which change will increase the solubility of a gas in water?
The solubility of a gas in a liquid will increase if: Lower the temperature of the solution , and therefore, lower the kinetic energy of the gaseous particles so they can escape the liquid phase less often.
How do temperature and pressure affect the solubility of gases in water?
Summary. The solubility of most substances depends strongly on the temperature and, in the case of gases, on the pressure. The solubility of most solid or liquid solutes increases with increasing temperature. The solubility of a gas decreases with increasing temperature.
Why does pressure increase gas solubility?
In general, the solubility of a gas in a liquid is increased by the increase of pressure. As the molecules collide more with the liquid’s surface, they will be able the squeeze between the liquid molecules and thus become a part of the solution.
What is the effect of increase in pressure on the solubility of a gas in a liquid Class 12?
Increase in pressure increases the solubility of a gas in a liquid.
How does pressure affect the solubility of a gas in a liquid quizlet?
At a given temperature the solubility of a gas in a liquid is directly proportional to the pressure of the gas above the liquid. As the pressure of the gas above the liquid increases, the solubility of the gas increases.
Why does increasing pressure increase gas solubility?
How does increasing the pressure of a solution affect the solubility of a solute in the solid state?
An increase in pressure and an increase in temperature in this reaction results in greater solubility. An increase in pressure results in more gas particles entering the liquid in order to decrease the partial pressure. Therefore, the solubility would increase.
How does the pressure of a gas over a liquid affect the quantity of gas that dissolves in solution?
Gases as might be expected, increase in solubility with an increase in pressure. Henry’s Law states that: The solubility of a gas in a liquid is directly proportional to the pressure of that gas above the surface of the solution. When the bottle is opened, the pressure above the solution decreases.
What are the factors that affect the solubility of a gas in a liquid?
Ans: The factors that affect the solubility of a gas in a liquid are the nature of the gas and solvent, temperature, and pressure.
Why does solubility increase with pressure?
How does pressure affect the solubility of a gas?
Gas solubility increases as the pressure of the gas increases. Carbonated beverages provide a nice illustration of this relationship. The carbonation process involves exposing the beverage to a relatively high pressure of carbon dioxide gas and then sealing the beverage container, thus saturating the beverage with CO 2 at this pressure.
What is the effect of temperature on solubility?
In the case of sparingly soluble substances which are liquid, the solubility increases with upsurge in temperature. In the case of gases, the solubility declines with a rise in temperature of the solvent. The bonds and forces between these molecules of solute are one of the factors affecting solubility.
What is the solubility of oxygen in water under pressure?
At 20 °C, the concentration of dissolved oxygen in water exposed to gaseous oxygen at a partial pressure of 101.3 kPa (760 torr) is 1.38 × 10 −3 mol L −1. Use Henry’s law to determine the solubility of oxygen when its partial pressure is 20.7 kPa (155 torr), the approximate pressure of oxygen in earth’s atmosphere.
How do you find the solubility of a gaseous solute?
Exposing a 100.0 mL sample of water at 0 °C to an atmosphere containing a gaseous solute at 20.26 kPa (152 torr) resulted in the dissolution of 1.45 × 10 −3 g of the solute. Use Henry’s law to determine the solubility of this gaseous solute when its pressure is 101.3 kPa (760 torr).