2,4-dinitrophenylhydrazine can be used as a method for identifying aldehydes and ketones – the 2,4-dinitrophenylhydrazone formed is a solid that can be purified and its melting point determined. Comparison of the melting point with a table of known values could identify the aldehyde or ketone.
How does aldehydes and ketones react with 2,4-DNP?
2,4-Dinitrophenylhydrazine: Aldehydes and ketones react with 2,4-dinitrophenylhydrazine reagent to form yellow, orange, or reddish-orange precipitates, whereas alcohols do not react. The mechanism of this reaction is that of imine formation and can be found in any organic lecture text.
What is the positive test for ketone with 2,4-DNP?
This solution is used to detect ketones and aldehydes. A positive test is signalled by the formation of a yellow, orange or red precipitate of the dinitrophenylhydrazone. Aromatic carbonyls give red precipitates whereas aliphatic carbonyls give more yellow color.
What happens when acetaldehyde treated with 2,4-DNP?
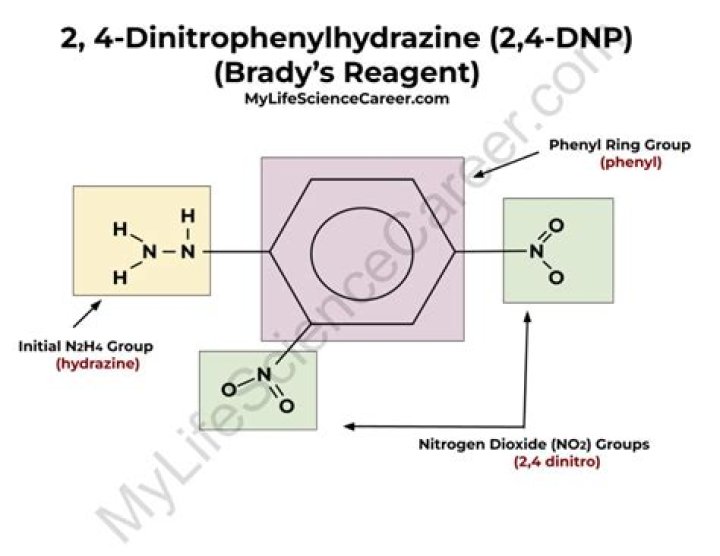
An aqueous solution of 2,4-dinitrophenyl hydrazine (DNP) is known as Brady’s reagent. It reacts with carbonyl compounds (aldehydes and ketone) to give a coloured precipitate. These precipitates have a sharp melting point. The melting points of the precipitates confirm the carbonyl compounds.
How do you test between aldehydes and ketones?
Tollens’ test, also known as silver-mirror test, is a qualitative laboratory test used to distinguish between an aldehyde and a ketone. It exploits the fact that aldehydes are readily oxidized (see oxidation), whereas ketones are not.
Is glucose give 2/4 DNP test?
Glucose does not gives 2,4−DNP test.
What does a negative 2,4-DNP test indicate?
A positive 2,4-DNP test means that your unknown contains either an aldehyde or ketone, while a negative test tells you that it doesn’t. It’s actually the hydrazine group that reacts with the aldehyde or ketone, producing a positive test.
What is the purpose of forming a derivative of a ketone using 2,4-DNP?
What is the purpose of forming a derivative of a ketone using 2,4-DNP. To identify unknown aldehydes and ketones that are liquid at room temperature.
Which reagent is used for test of aldehyde and ketone?
Once the presence of a carbonyl group has been identified using 2,4-dinitrophenylhydrazine (also known as Brady’s reagent or 2,4-DNPH or 2,4-DNP), Tollens’ reagent can be used to distinguish ketone vs aldehyde.
What is the general test for the presence of aldehydes?
Tollen’s reagent
Tollen’s reagent is a classical organic laboratory technique to test for the presence of an aldehyde. The reagent consists of silver(I) ions dissolved in dilute ammonia. When the aldehyde is oxidized, the silver(I) ions are reduced to silver metal.
Which does not give 2/4 DNP test?
Glucose does not gives 2,4 – DNP test.
What is the 2-4-dinitrophenylhydrazine test for aldehydes?
Positive (with precipitate) and negative result with the 2,4-Dinitrophenylhydrazine test for aldehydes and ketones. Photo by Sonjiala Hotchkiss/STEM Punk Both aldehydes and ketones have a carbonyl group (a carbon double bonded to oxygen).
How to distinguish aldehyde from ketone in Class 12 chemistry?
Keep visiting BYJU’S to learn more about class 12 CBSE chemistry practicals. Name any one test to distinguish aldehyde from ketone. Tollens ‘ test is a qualitative laboratory test used to distinguish between an aldehyde and a ketone, also known as a silver-mirror test.
How to test for the presence of allylic alcohol in ketones?
The 2,4-dinitrophenylhydrazine reagent will already be prepared for you. Formation of a precipitate is a positive test. Some ketones give oils which will not solidify. Some allylic alcohols are oxidized by the reagent to aldehydes and give a positive test.
What reagent gives a positive test for aldehydes?
Aldehydes. A positive test is marked by the formation of a green color within 5 seconds upon addition of the orange-yellow reagent to a primary or secondary alcohol. Aldehydes also give a positive test, but tertiary alcohols do not. The Jones reagent will already be prepared for you.